Allergan Partners with Urine Incontinence Treatment Company
Allergan is conducting clinical trials for a Botox-based bladder treatment, using a sustained release gel developed by Israel-based UroGen
12:2308.11.17
Allergan Pharmaceuticals International Ltd., a wholly owned subsidiary of Allergan PLC, has started a phase 2 clinical trial for the treatment of overactive bladder. The trial will combine Botox with a sustained release gel called RTGel, developed by Nasdaq-listed UroGen Pharma Ltd., a clinical stage company developing non-surgical urology treatments.
Related articles
RTGel is a temperature-sensitive substance that changes from a liquid to a gel when it reaches body temperature. These properties enable a slower release of drugs in the body when mixed with the gel. Botox is already in use for treating overactive bladder, since it relaxes the nerves that over-stimulate the bladder muscles and lead to frequent urination.
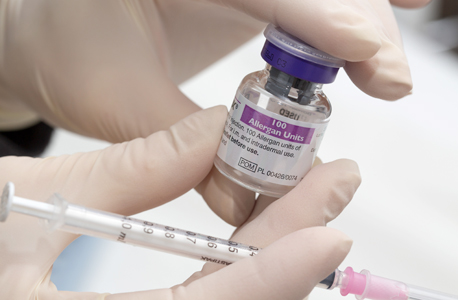
Allergan's Botoxצילום: בלומברג
The current trial will enroll up to 335 patients with overactive bladder who have an unsatisfactory response to current pharmacologic treatments. Patients will receive a single RTGel-Botox bladder instillation. Bladder instillation is the process of introducing drugs into the bladder using a catheter under hospital monitoring.
Israel-based UroGen first announced the collaboration with Allergan in October 2016, when Allergan licensed the worldwide rights for RTGel. As part of the agreement, Allergan made an upfront payment of $17.5 million as part of the deal. UroGen is eligible to receive up to $200 million in additional payments following certain milestones, and is also entitled royalties.
In July, UroGel received $7.5 million from Allergan after the latter submitted an application for RTGel to the U.S. Food and Drug Administration, for a specific Botox-based bladder treatment. The gel was designed to work with multiple active pharmaceutical ingredients.
UroGel’s stock has risen by 165.7% since the company’s Nasdaq initial public offering in May. Arie Belldegrun, the founder of cancer treatment company Kite Pharma recently sold to Gilead Sciences Inc. for $11.9 billion, sits as UroGel’s chairman of the board.



