Why an early Tesla investor is betting big on Israeli brain technology
BrainsWay’s FDA approvals and clinic expansion strategy are attracting global capital.
For Antonio Gracias, founder of the American private equity fund Valor Equity Partners, the first institutional investor in Tesla, his investment in the Israeli medical device company BrainsWay since the end of 2024 has generated a return 3.5 times higher than that achieved by Tesla shares over the same period. This comes after BrainsWay reached a record valuation of NIS 1.6 billion (approximately $515 million) before dropping to its current market cap of NIS 1.5 billion ($485M).
In September 2024, Valor, which manages $17.5 billion in assets and specializes in investments in technology companies, acquired 14% of BrainsWay for $20 million, becoming its largest shareholder. Gracias himself also became a private investor and now holds 1.4% of the company’s shares.
Valor no longer holds Tesla shares, although Gracias continues to hold them personally. Since becoming an interested party in BrainsWay in November 2024, his holding has generated a return of 98%, compared with a rise of only 28% in Tesla shares during the same period.
The surge in BrainsWay’s stock did not begin with Valor’s entry, but rather continued a trend that began nearly a year and a half earlier, around the end of the first half of 2023, near the time CEO Hadar Levy took office. From a valuation of NIS 92 million in May 2023, the company climbed to a record NIS 1.6 billion last month, a cumulative return of 1,288.6% in just over three and a half years. For comparison, the Tel Aviv-125 index rose by 133.2% during the same period.
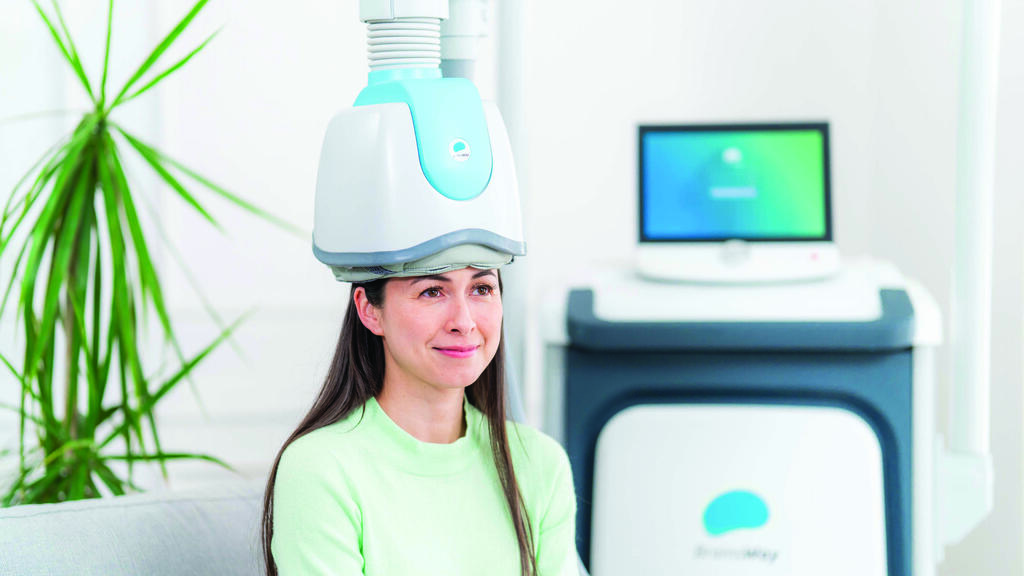
BrainsWay, which develops medical devices for the non-invasive treatment of brain disorders, primarily mental illnesses and addictions, is listed on both the Tel Aviv Stock Exchange and Nasdaq. Its technology is based on deep transcranial magnetic stimulation (Deep TMS). The treatment is delivered through a helmet-like device placed on the patient’s head that transmits magnetic pulses according to a prescribed treatment protocol.
Last September, the company received approval from the US Food and Drug Administration (FDA) allowing physicians to provide patients suffering from major depression with an accelerated treatment protocol using its system: just six days of treatment, with five sessions per day. Previously, the protocol required five weekly treatments over four weeks.
After a long period of losses, BrainsWay returned to net profitability in the fourth quarter of 2023 and has maintained a positive bottom line since then. The year 2024 was the first profitable full year in the company’s history.
In each of the last six quarters, through the third quarter of 2025, the company recorded revenue growth of 26%-29% compared with the corresponding quarter a year earlier. BrainsWay expects to end 2025 with revenue of $52 million, compared with $41 million in 2024, an increase of 27%, and with an operating margin of 6%-7%, compared with 3.4% in 2024. The company expects similar growth rates in 2026.
In a conversation with Calcalist, Levy said: “We are managing to combine advanced technology with two critical commercial parameters for a medical device company: FDA approval of the treatment we offer and the ability to contract with insurance companies and receive reimbursement from them, processes that require long-term effort.”
According to Levy, “The United States is currently our main market, accounting for 85% of our revenue. It is a market of 250 million insured individuals covered by insurance companies that reimburse our treatment. For patients, the abbreviated treatment increases the likelihood that they will complete the protocol, and its financial cost is lower thanks to reimbursement and a simplified approval process.”
Insurance companies provide reimbursement of about $8,500 per patient, while potentially saving costs estimated at $40,000-$50,000, the approximate cost of alternative drug treatments that often require long adjustment periods and are not always successful. At the same time, the reimbursement is shared between BrainsWay and the treating clinic or physician. The abbreviated treatment protocol also enables earlier reimbursement.
BrainsWay’s systems are currently operating in approximately 1,700 clinics across the United States. Levy notes that “we are constantly working to expand into additional clinics, mainly through clinic chains, in order to grow without significantly increasing marketing expenses.”
“In addition, we have implemented a fundamental change in the business model: in the past we sold the system in a one-time transaction, whereas today contracts are long-term and customers pay for usage rather than the system itself. Ninety-three percent of customers are repeat customers, either under fixed-price contracts or a pay-as-you-go model. We can monitor the scope of usage, and when we identify activity levels that are lower than expected, we contact the clinic, analyze the reasons together, and look for solutions.”
BrainsWay’s treatment is intended for patients suffering from treatment-resistant major depression, a combination of depression and anxiety, obsessive-compulsive disorder (OCD), and smoking addiction.
“We are also exploring additional growth engines,” says Levy. “We are conducting trials to examine the effectiveness of the treatment for patients suffering from post-traumatic stress disorder and are also aiming to expand into the treatment of alcohol addiction.”
Regarding PTSD, the Rehabilitation Division of Israel’s Ministry of Defense approved reimbursement in September 2024 for treatment using BrainsWay’s technology for patients suffering from the disorder.
However, sales volumes in Israel remain relatively negligible. According to Levy, “Our treatment is included in the national health basket, and the systems operate in the major hospitals. We are currently in discussions with the health funds to expand the treatment into community clinics as well.”
Another direction the company began pursuing in 2025 is investing, as a minority partner, in clinic chains, typically with investments totaling several million dollars.
Last week, for example, the company signed an agreement with a Canadian mental health clinic chain for an investment that could reach $2.5 million.
According to Levy, “This is an expansion strategy we formulated together with Valor. Through it, we are using the $71 million in cash we have accumulated to accelerate organic growth. We are investing $1.5-3 million in clinic chains that are seeking to expand and open additional locations. This makes it easier for us to integrate our systems and grow alongside them. So far we have invested in four chains, and the goal for 2026 is to invest in ten more.”
Despite the company’s strong momentum, the industry in which it operates is not without risks.
“We must move quickly,” says Levy. “As our technology advances, another technology in the mental health field could emerge that changes the market and that insurance companies might prefer.”
Accordingly, in 2025 BrainsWay announced a strategic investment in the Israeli company Neurolief, which develops a home treatment system for major depression. BrainsWay granted Neurolief a convertible loan of $5 million, with an option to acquire the company in the future.
“We realized that Neurolief is, on the one hand, a competitor, but on the other hand it provides a complementary treatment,” Levy explains. “The United States is a huge market, and not every patient can or wants to come to a clinic. The combination of the two systems will allow us to reach a wider audience, and eventually we may combine the treatments.”